3D Culture

Cells used in pre-clinical research are traditionally cultured on a 2-dimensional surface, typically on the bottom of a plastic container. Even for a non-cell biologist, like me, it is easy to understand that this is very far from a natural cell environment. In our bodies, cells live in a protein gel similar to the gel created by using ordinary cooking gelatin. Cell biologists call this gel the extracellular matrix.
3D cell culture
To make experiments in pre-clinical research more realistic and to reduce animal testing, researchers are increasingly culturing cells embedded in a 3-dimensional layer of collagen – the main structural protein of the extracellular matrix. Conventional microscopy has a very limited focal depth of just a few micrometers. This makes it difficult to create focused images of the cells, as they move out of focus when they move about in the 3-dimensional gel.
Digital autofocus
In contract to conventional microscopy, our HoloMonitor® instrument digitally focuses the image after it is recorded. This enables HoloMonitor to create sharp images of the cells wherever they are located in the gel.


AACR-NCI-EORTC poster
On the 6th of November researchers from the Holographic Imaging Center of Excellence at Northeastern University in Boston presented a poster at the AACR-NCI-EORTC International Conference on Molecular Targets and Cancer Therapeutics.
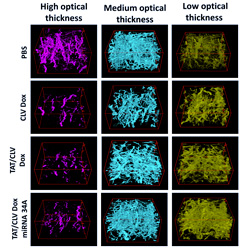
Using HoloMonitor and a public domain image processing software called Image J, the researchers from Northeastern University have developed a method for cell experiments based on cells cultured in a 3-dimensional collagen gel. They show that this new method is clearly superior to conventional methods, based on cells cultured on a 2-dimensional surface, and conclude:
“We developed a novel holographic imaging method using HoloMonitor and Image J. The new method is an important step in developing methods that better emulate biological processes and offer the possibility of evaluating effects of drugs at lower cost and experimental complexity than methods based on animals.”— Ed Luther et al
Northeastern University