Interim Report 3 2024/25
Phase Holographic Imaging PHI AB (publ)
Lund, March 13, 2025
November 2024 – January 2025
| Net sales | 6 481 (4 873) KSEK |
| Operating result before depreciation (EBITDA) | -987 (-3 179) KSEK |
| Net result | -2 681 (-5 152) KSEK |
| Earnings per share | -0.08 (-0.21) SEK |
| Gross margin | 76 (72) % |
May 2024 – January 2025
| Net sales | 10 796 (8 824) KSEK |
| Operating result before depreciation (EBITDA) | -8 510 (-9 568) KSEK |
| Net result | -12 280 (-14 826) KSEK |
| Earnings per share | -0.34 (-0.58) SEK |
| Gross margin | 71 (73) % |
In Short
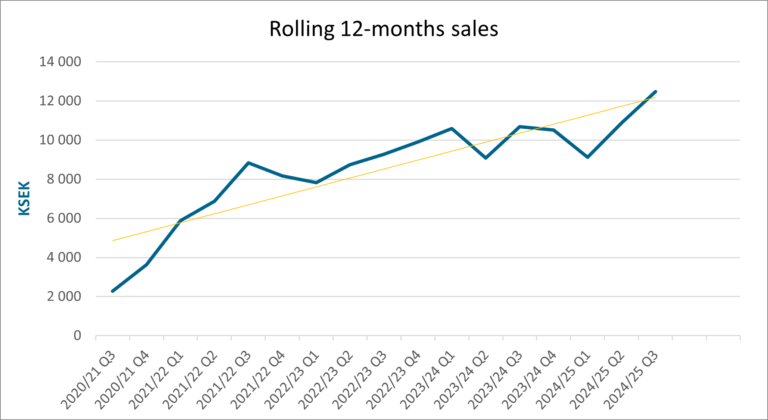
- Sales grew significantly for both Q3 and YTD, outperforming the FY results of the previous year.
- Gross margin continues to be strong, even slightly above planned levels (+4pp).
- Overall costs remain stable and as per plan; higher admin costs (capital raise connected) vs. lower R&D expenses.
- The absolute key to transforming the Company, both short-term and long-term, is a significant increase in sales, which is expected due to a strong pipeline & company potential.
CEO Commentary
Having recently taken on the role of acting CEO at PHI, my primary focus is execution—translating the Company’s potential into tangible results. We have a strong foundation built on deep technical expertise and a clear commitment to advancing regenerative medicine. Now, it’s time to maximize that potential.
Our path forward is clear: be the best at what we know best. This means focusing on cutting-edge imaging technology that empowers cell research and therapy, supported by a strong distributor network and strategic partnerships. Our recent financial performance demonstrates that we are moving in the right direction, with growing sales and a strong gross margin. However, we must accelerate further, ensuring our innovations reach the market at scale.
With a committed team, solid industry relationships, and a clear mission, we are ready to take PHI to the next level. The focus now is on execution and delivering on our promises—because the opportunity ahead is too great not to seize.

About PHI
Quantitative Phase Imaging
Phase Holographic Imaging (PHI), a leading medical technology company, develops and markets its non-invasive time-lapse imaging instruments for studying living cells.
The foundation of PHI’s current commercial HoloMonitor® products is Quantitative Phase Imaging (QPI) technology. This technology brings an innovative approach to real-time cell quality evaluation. It offers a detailed analysis of a large number of cell health and behavior characteristics without harming or influencing living cells and thus differing from conventional measurement methods, which often jeopardize cell integrity.

The addressable market
PHI has been active within the pre-clinical and biomedical cell research market, having established a global presence with HoloMonitor® systems and their scientific validation in both academia and industry, primarily addressing cancer, stem cell and drug development research.
PHI’s HoloMonitor technology is transforming pre-clinical research by providing a foundation for better cell models, which are critical before clinical drug testing. This innovative approach, utilizing Quantitative Phase Imaging (QPI), ensures cells remain unaffected during analysis. Unlike conventional cell measurement methods that often require genetic manipulation or staining with toxic substances, QPI offers a non-invasive alternative that maintains the integrity of cell cultures. This advancement addresses the critical need for accurate, cost-efficient pre-clinical data to reduce the high failure rates in drug development. With 9 out of 10 drugs failing in clinical trials due to ineffective results or adverse effects, largely stemming from flawed pre-clinical outcomes, PHI’s technology represents a significant leap forward. By enabling scientists to obtain better data without compromising cell health, HoloMonitor sets a new standard in drug development and basic medical research, aiming for more successful patient outcomes and reduced research expenses.
PHI is strategically prepared to extend its reach into the large clinical market and emerging regenerative medicine field, which presents significant growth opportunities. By striving to achieve Good Manufacturing Practice (GMP) standards and create a company quality management system (QMS), PHI aims to penetrate these markets in the future, where its non-invasive cell analysis solutions can offer critical cell quality control assessment.
Regenerative medicine is a groundbreaking field focused on developing methods to regenerate, repair, or replace damaged cells, tissues, or organs. It integrates biology, chemistry, computer science, and engineering to develop treatments for conditions previously thought untreatable. It has already begun to transform healthcare by offering new hope to patients with conditions like cancer, Parkinson’s disease, diabetes, and deafness, displaying its vast potential to improve and save lives around the globe.
Business model & strategy
PHI markets a competitive combination of sophisticated software and quality hardware, constantly evolving its offerings through in-house development in response to changing market needs. The production of the hardware and accessories is strategically outsourced to specialized subcontractors located in Sweden.
PHI’s business model strategically centers around the HoloMonitor portfolio, designed to meet the specific needs of academia and industry research labs. The Company prioritizes direct interactions and live product demonstrations at potential customer facilities, particularly in cell-based research environments. Combined with a digital marketing approach, this sales strategy is essential for demonstrating HoloMonitor’s advantages firsthand, aiming to foster trust and enabling customers to assess its suitability for their unique research needs. The sales process at PHI is designed to align with the detailed and extended decision-making cycles often found within the scientific community. It accounts for the crucial phase of securing grant funding or other financial support for institutions looking to invest in new research equipment. As a result, sales cycles typically span 6 to 12 months.
PHI is actively expanding its global reach within its current core markets, such as North America, Europe, Asia, and Australia, while also exploring new territories. At the heart of this strategic expansion is the partnership with Altium, a major investor and global distributor of PHI. Altium’s engagement has been transformative for PHI’s operations, not only providing substantial financial backing but also significantly enhancing PHI’s market presence through Altium’s well-established sales channels and distribution networks. The joint efforts aim to broaden the geographic reach and adoption of the HoloMonitor system. The efforts utilize targeted demonstrations and deeper market penetration to enhance sales performance and cash flow, positioning PHI as a key player in its field. This collaboration enables PHI to focus on its core expertise and innovation capabilities—advancing QPI technology and product development within regenerative medicine.
Achieving future goals
PHI is focusing on achieving important milestones in the coming years, utilizing the potential of its partnerships and collaborations. PHI has a clear focus on expanding its business to the clinical research market and positioning itself in the Regenerative Medicine field.
The collaboration with Altium is instrumental in PHI’s approach to gaining a stronger foothold in the regenerative medicine field, where PHI’s cell quality control technology can meet critical industry needs. PHI is also actively fostering alliances with leading institutions such as the Wake Forest Institute for Regenerative Medicine (WFIRM), renowned for its groundbreaking contributions to translating scientific research into clinical therapies. Collaborating with WFIRM enriches PHI’s initiatives, providing access to an exceptional ecosystem of expertise and innovation. This partnership bolsters PHI’s leadership in regenerative medicine, enabling the ongoing development and application of PHI’s cell quality control technology.
PHI engages in these strategic relationships to fortify the Company’s position as a thought leader in the field with the intention to accelerate progress, bolster market penetration, open new doors to new business opportunities, and enhance shareholder value.
Net Sales and Result
Net sales for the third quarter amounted to 6 481 (4 873) KSEK. Operating results before depreciation (EBITDA) amounted to -987 (-3 179) KSEK for the third quarter. The net result for the third quarter amounted to -2 681 (-5 152) KSEK.
INVESTMENTS
The company invested 1 813 (2 805) KSEK in the product, patent, and application development during the third quarter.
FINANCING
Cash, cash equivalents, and unutilized granted credits amounted to 26 567 (684) KSEK by the end of the period. The equity ratio was 85 (30) %. As was announced in November 2024, the company secured a rights issue where the proceeds amounted to SEK 64.3 million before deduction of transaction costs and offsetting of loans, which were received in January.
The Rights Issue has been registered with the Swedish Companies Registration Office. The total number of shares has increased by 35,729,342 shares, from 27,322,438 shares to 63,051,780 shares, and the Company’s share capital has increased by SEK 7,145,868.40, from SEK 5,464,487.60 to SEK 12,610,356.00.
RISKS
The company may be affected by various factors described in the Annual Report. These factors may individually or jointly increase risks for the operation and result of the company.
ACCOUNTING PRINCIPLES
The accounts are prepared in accordance with the Annual Accounts Act and general advice from the Swedish Accounting Standards Board BFNAR 2012:1 Annual accounts and consolidated accounts (K3).
REVIEW
This interim report has not been subject to review by the company’s auditor.
STATEMENTS ABOUT THE FUTURE
In this report, statements concerning the company’s business environment and future reflect the board of directors’ current view of future events and financial developments. Forward-looking statements only express the judgments and assumptions made by the board of directors on the day of the report. These statements have been carefully assessed. However, it is brought to the reader’s attention that these statements are associated with uncertainty, like all statements about the future.
CALENDAR
June 26, 2025, Interim Report 4 2024/2025
On behalf of the Board of Directors
Ivan Jurkovic, CEO
For additional information, please contact:
E-mail: ir@phiab.com
Web: www.phiab.com
Consolidated – PHI Group
Income statement (KSEK)
| Q3 | Q3 | YTD | YTD | FY | |
| 2024/25 | 2023/24 | 2024/25 | 2023/24 | 2023/24 | |
| Net sales | 6 481 | 4 873 | 10 796 | 8 842 | 10 510 |
| Cost of products sold | -1 573 | -1 374 | -3 156 | -2 390 | -3 071 |
| Gross profit | 4 907 | 3 499 | 7 639 | 6 434 | 7 439 |
| Gross margin | 76% | 72% | 71% | 73% | 71% |
| Selling expenses | -1 907 | -2 848 | -6 797 | -8 243 | -10 898 |
| Administrative expenses | -3 137 | -1 590 | -6 293 | -3 379 | -5 930 |
| R&D expenses | -1 240 | -2 363 | -4 228 | -6 244 | -7 554 |
| Operating result (EBIT) | -1 377 | -3 302 | -9 679 | -11 432 | -16 943 |
| Financial net | -1 304 | -1 850 | -2 601 | -3 394 | -3 232 |
| Result before tax (EBT) | -2 681 | -5 152 | -12 280 | -14 826 | -20 175 |
| Net Result (EAT) | -2 681 | -5 152 | -12 280 | -14 826 | -20 175 |
Balance sheet (KSEK)
| YTD | YTD | FY | |
| 2024/25 | 2023/24 | 2023/24 | |
| ASSETS | |||
| Non-current assets | |||
| Intangible assets | 30 852 | 24 202 | 25 565 |
| Tangible assets | 1 123 | 613 | 1 011 |
| Financial assets | 728 | 153 | |
| Total non-current assets | 31 975 | 24 815 | 26 729 |
| Current Assets | |||
| Inventory | 2 102 | 4 752 | 3 732 |
| Short-term receivables | 2 906 | 5 638 | 2 374 |
| Cash and equivalents | 24 567 | 21 | 167 |
| Total current assets | 29 576 | 10 411 | 6 273 |
| Total assets | 61 551 | 35 954 | 33 002 |
| EQUITY AND LIABILITIES | |||
| Equity | 52 047 | 10 892 | 4 944 |
| Financial liabilities | 830 | 18 191 | 17 845 |
| Operating liabilities | 8 675 | 6 871 | 10 213 |
| Total equity and liabilities | 61 551 | 35 954 | 33 002 |
Changes in equity (KSEK)
| YTD | YTD | FY | |
| 2024/25 | 2023/24 | 2023/24 | |
| Opening Balance | 4 988 | 3 728 | 3 728 |
| Equity issues, net | 59 338 | 22 617 | 22 617 |
| Net profit | -12 280 | -14 826 | -20 175 |
| Translation difference | -628 | -1 182 | |
| Closing balance | 52 047 | 10 892 | 4 988 |
| Equity ratio | 84.6% | 30.3% | 15.0% |
Cash flow analysis (KSEK)
| Q3 | Q3 | YTD | YTD | FY | |
| 2024/25 | 2023/24 | 2024/25 | 2023/24 | 2023/24 | |
| Operating activities | |||||
| Net result | -2 681 | -5 152 | -12 280 | -9 675 | -20 175 |
| Depreciation | 390 | 876 | 1 169 | 1 742 | 3 500 |
| Translation difference | -277 | 296 | -221 | 140 | -634 |
| Operating cash flow | -2 569 | -3 980 | -11 332 | -7 793 | -17 309 |
| Increase (-)/decrease (+) in inventories | 212 | 180 | 1 630 | -1 086 | 114 |
| Increase (-)/decrease (+) in operating receivables | -708 | -2 198 | -532 | 1 982 | 2 919 |
| Increase (+)/decrease (-) in operating liabilities | -15 357 | -1 625 | -1 494 | -2 033 | -2 483 |
| Change in working capital | -15 853 | -3 643 | -397 | -1 137 | 550 |
| Cash flow from operating activities | -18 422 | -7 623 | -11 729 | -8 930 | -16 759 |
| Investing activities | |||||
| Development expenses | -1 813 | -2 805 | -6 194 | -4 249 | -9 056 |
| Patents | -25 | 25 | -110 | ||
| Tangible assets | -146 | -601 | |||
| Financial fixed assets | -153 | ||||
| Cash flow after investments | -20 235 | -10 599 | -17 923 | -13 153 | -26 679 |
| Financing activities | |||||
| Net proceeds from equity issues | 54 183 | 10 497 | 59 337 | 11 492 | 22 023 |
| Increase (+)/decrease (-) in borrowings | -17 300 | -17 015 | -3 524 | -2 222 | |
| Utilized credits | 1 737 | ||||
| Cash flow from financing activities | 36 883 | 10 497 | 42 322 | 7 968 | 21 538 |
| Cash flow for the period | 16 648 | -101 | 24 400 | -5 185 | -5 141 |
| Cash and cash equivalents at the beginning of the period | 7 919 | 122 | 168 | 5 307 | 5 308 |
| Cash and cash equivalents at the end of the period | 24 567 | 21 | 24 567 | 122 | 167 |
| Incl. unutilized credits | 26 567 | 664 | 26 567 | 787 | 429 |
Data per share
| Q3 | Q3 | YTD | YTD | FY | |
| 2024/25 | 2023/24 | 2024/25 | 2023/24 | 2023/24 | |
| Earnings per Share, SEK | -0,08 | -0,21 | -0,34 | -0,58 | -0,08 |
| Equity per share, SEK | 0,83 | 0,42 | 0,83 | 0,42 | 0,19 |
| Number of Shares, end of period | 63 051 780 | 26 192 925 | 63 051 780 | 26 192 925 | 26 192 925 |
| Average number of shares | 34 165 202 | 24 354 950 | 36 344 443 | 25 495 966 | 24 808 150 |
| Share price end of period, SEK | 1.22 | 6.46 | 1.22 | 6.46 | 7.80 |
Parent company
Income statement (KSEK)
| Q3 | Q3 | YTD | YTD | FY | |
| 2024/25 | 2023/24 | 2024/25 | 2023/24 | 2023/24 | |
| Net sales | 6 159 | 4 847 | 10 418 | 7 886 | 9 657 |
| Cost of products sold | -1 463 | -1 637 | -3 033 | -2 647 | -3 506 |
| Gross profit | 4 696 | 3 210 | 7 385 | 5 239 | 6 151 |
| Gross margin | 76% | 66% | 71% | 66% | 64% |
| Selling expenses | -794 | -1 668 | -3 273 | -4 498 | -5 877 |
| Administrative expenses | -3 137 | -1 590 | -6 293 | -3 379 | -5 929 |
| R&D expenses | -1 240 | -2 363 | -4 228 | -6 244 | -7 554 |
| Operating result (EBIT) | -476 | -2 411 | -6 408 | -8 882 | -13 209 |
| Financial net | -1 304 | -1 850 | -2 601 | -3 394 | -3 232 |
| Result before tax (EBT) | -1 780 | -4 261 | -9 010 | -12 276 | -16 441 |
| Net Result (EAT) | -1 780 | -4 261 | -9 010 | -12 276 | -16 441 |
Balance sheet (KSEK)
| YTD | YTD | FY | |
| 2024/25 | 2023/24 | 2023/24 | |
| ASSETS | |||
| Non-current assets | |||
| Intangible assets | 28 097 | 23 069 | 23 986 |
| Tangible assets | 1 123 | 613 | 1 011 |
| Financial assets | 17 789 | 12 066 | 14 003 |
| Total non-current assets | 47 009 | 35 748 | 39 000 |
| Current Assets | |||
| Inventory | 1 206 | 3 962 | 2 734 |
| Short-term receivables | 2 709 | 5 565 | 2 242 |
| Cash and equivalents | 24 331 | 90 | |
| Total current assets | 28 246 | 9 527 | 5 066 |
| Total assets | 75 256 | 45 275 | 44 066 |
| EQUITY AND LIABILITIES | |||
| Equity | 66 625 | 20 265 | 16 100 |
| Financial liabilities | 18 191 | 17 841 | |
| Operating liabilities | 8 630 | 6 819 | 10 125 |
| Total equity and liabilities | 75 256 | 45 275 | 44 066 |